Copper(II) fluoride or cupric fluoride is an inorganic compound with the chemical formula CuF2. The anhydrous form is a white, ionic, crystalline, hygroscopic salt with a distorted rutile-type crystal structure, similar to other fluorides of chemical formulae MF2 (where M is a metal). The dihydrate, CuF2·2H2O, is blue in colour.[6]
Properties
Copper(II) fluoride is slightly soluble in water, but starts to decompose in hot water, producing basic F− and Cu(OH)+ ions.[citation needed]
Structure
Copper(II) fluoride has a monoclinic crystal structure[7] and cannot achieve a higher-symmetry structure. It forms rectangular prisms with a parallelogram base.[8] Each copper ion has four neighbouring fluoride ions at 1.93 Å separation and two further away at 2.27 Å.[6] This distorted octahedral [4+2] coordination is a consequence of the Jahn–Teller effect in d9 copper(II),[9] and leads to a distorted rutile structure similar to that of chromium(II) fluoride, CrF2, which is a d4 compound.[6]
| Copper coordination | Fluorine coordination |
|---|---|
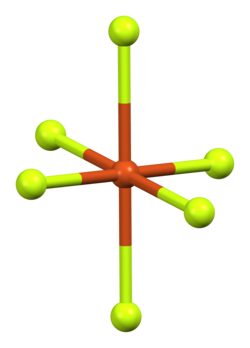
|

|
Synthesis
Copper(II) fluoride can be synthesized from copper and fluorine at elevated temperatures. At 500 °C (932 °F) a 53% conversion is achieved.[10]
- Cu + F2 → CuF2
Uses
Cupric fluoride catalyzes the decomposition of nitric oxides in emission control systems.[11]
Copper(II) fluoride can be used to make fluorinated aromatic hydrocarbons by reacting with aromatic hydrocarbons in an oxygen-containing atmosphere at temperatures above 450 °C (842 °F). This reaction is simpler than the Sandmeyer reaction, but is only effective in making compounds that can survive at the temperature used. A coupled reaction using oxygen and 2 HF regenerates the copper(II) fluoride, producing water.[12] This method has been proposed as a “greener” method of producing fluoroaromatics since it avoids producing toxic waste products such as ammonium fluoride.
Reactions
It loses fluorine in the molten stage at temperatures above 950 °C (1742 °F).[citation needed]
- 2 CuF2 → 2 CuF + F2
- 2 CuF → CuF2 + Cu
The complex anions CuF3−, CuF42− and CuF64− are formed if CuF2 is exposed to substances containing fluoride ions F−.[citation needed]
References
- ^ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ^ a b T. C. Ehlert; J. S. Wang (1977). “Thermochemistry of the copper fluorides”. Journal of Physical Chemistry. 81 (22): 2069–2073. doi:10.1021/j100537a005.
- ^ Kent, R. A.; Mcdonald, J. D.; Margrave, J. L. (1966). “Mass Spectrometric Studies at High Temperatures. IX. The Sublimation Pressure of Copper(II) Fluoride”. Journal of Physical Chemistry. 70 (3): 874–877. doi:10.1021/j100875a042.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. “#0150”. National Institute for Occupational Safety and Health (NIOSH).
- ^ “Copper Fluoride – ESPI Metals”. www.espimetals.com. Retrieved 2026-02-19.
- ^ a b c d Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1184–1185. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ a b Fischer, P.; Hälg, W.; Schwarzenbach, D.; Gamsjäger, H. (1974). “Magnetic and crystal structure of copper(II) fluoride”. J. Phys. Chem. Solids. 35 (12): 1683–1689. doi:10.1016/S0022-3697(74)80182-4.
- ^ C. Billy; H. M. Haendler (1957). “The Crystal Structure of Copper(II) Fluoride”. Journal of the American Chemical Society. 79 (5): 1049–51. doi:10.1021/ja01562a011.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1190–1191. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ H. M. Haendler; L. H. Towle; E. F. Bennett; W. L. Patterson (1954). “The Reaction of Fluorine with Copper and Some of its Compounds. Some Properties of Copper(II) Fluoride”. Journal of the American Chemical Society. 76 (8): 2178–2179. doi:10.1021/ja01637a039.
- ^ Meshri, Dayal T. (2000), “Fluorine compounds, inorganic, copper”, Kirk-Othmer Encyclopedia of Chemical Technology, New York: John Wiley, doi:10.1002/0471238961.0315161613051908.a01, ISBN 9780471238966
- ^ M. A. Subramanian; L. E. Manzer (2002). “A “Greener” Synthetic Route for Fluoroaromatics via Copper (II) Fluoride”. Science. 297 (5587): 1665. doi:10.1126/science.1076397. PMID 12215637. S2CID 32697750.
Further reading
- Shashkin, S. Y.; Goddard III, W. A. (1986). “Electron Correlation effects in ligand field parameters and other properties of copper II fluoride”. Journal of Physical Chemistry. 90 (2): 250–255. doi:10.1021/j100274a010.
- P. C. de Mello; M. Hehenberg; S. Larson; M. Zerner (1980). “Studies of the electronic structure of copper fluorides and copper chlorides”. Journal of the American Chemical Society. 102 (4): 1278–1288. doi:10.1021/ja00524a010.
