Renal artery stenosis (RAS) is the narrowing of one or more of the renal arteries, which can reduce kidney blood flow and may lead to hypertension and kidney disease. RAS is often caused by atherosclerosis (approximately 90%) which is related to hypertension, high cholesterol, and smoking, or other rarer disease processes such as fibromuscular dysplasia (approximately 10%)[4]. The narrowing of one or more renal arteries can impede blood flow to a kidney, which the kidney can interpret as hypoperfusion (decreased or inadequate blood flow to an organ or tissue), and through a cascade of events involving activation of the Renin Angiotensin Aldosterone System (RAAS) may result in renovascular hypertension – a secondary type of high blood pressure.[5] Possible complications of renal artery stenosis are chronic kidney disease, kidney atrophy, resistant hypertension, cardiovascular events such as coronary artery disease and stroke, and fluid retention, often manifesting as lower extremity edema.[1][6]
Signs and symptoms
Most cases of renal artery stenosis are asymptomatic, but may manifest with signs of fluid retention, especially lower extremity edema. The main complication of RAS is resistant hypertension, or high blood pressure that cannot be controlled despite the concurrent use of three or more different classes of antihypertensive medications.[7][8] Chronic kidney disease may develop if one or both kidneys do not receive adequate blood flow over time. A rare complication of RAS is flash pulmonary edema (prevalence of 10-30% of patients), often referred to as Pickering Syndrome.[9][10] This complication occurs more commonly in those with bilateral RAS, and is characterized by the rapid accumulation of fluid in the lungs and possibly leading to respiratory failure.
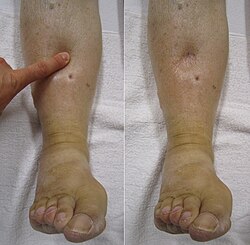
Pathophysiology
As stated above, renal artery stenosis is most often caused by atherosclerosis, which causes the renal arteries to harden and narrow due to the build-up of plaque. This is known as atherosclerotic renovascular disease, which accounts for about 90% of cases.[11] This narrowing of renal arteries decreases blood flow to the kidney. As less blood flows through the kidneys, specialized cells within the walls of the afferent arterioles of the kidney called Juxtaglomerular cells act as miniature stretch receptors to sense this low blood pressure and secrete the enzyme Renin, which begins the activation of the Renin Angiotensin Aldosterone System (RAAS). The liver produces the precursor angiotensinogen, which is converted into angiotensin I by renin (secreted by the kidneys). Angiotensin I is then converted into the active hormone, angiotensin II, primarily by angiotensin-converting enzyme (ACE), largely located in the lungs. Angiotensin II exerts widespread vasoconstriction and triggers the release of Aldosterone, which essentially prompts the kidneys to retain sodium and water, ultimately leading to increased blood pressure. Therefore, since people with RAS have chronic narrowing of their renal arteries, they may have chronic resistant high blood pressure because their RAAS is constantly activated.[12]


Pathophysiological sequelae
The pathophysiology of renal artery stenosis leads to changes in the structure of the kidney that are most noticeable in the tubular tissue.[13]
Changes include:[13]
- Fibrosis
- Tubular cell size (decrease)
- Thickening of Bowman capsule
- Tubulosclerosis
- Glomerular capillary tuft (atrophy)
Diagnosis
A variety of techniques along with a clinical prediction rule can be used to help guide the diagnosis of RAS.[14] Patients may present to a cardiologist or nephrologist with resistant high blood pressure, who may start with a renal ultrasound (US) to look for signs of possible RAS, such as atrophy or a shrunken kidney due to chronic hypoperfusion. [15] Although less sensitive, physical exam with auscultation using a stethoscope may reveal a renal bruit. Following US, confirmatory testing is usually done via a doppler or angiography, in part depending on if the patient has adequate kidney function to tolerate the contrast used in angiography.[16]
Among the diagnostic techniques are:
- Doppler ultrasound study of the kidneys[2]
- Refractory hypertension[17]
- Auscultation (with stethoscope) – bruit (“rushing” sound)[18]
- Captopril challenge test[3]
- Captopril test dose effect on the differential renal function as measured by MAG3 scan.[19]
- Renal artery arteriogram.[20][21]
The specific criteria for renal artery stenosis on Doppler are an acceleration time of greater than 70 milliseconds, an acceleration index of less than 300 cm/sec² and a velocity ratio of the renal artery to aorta of greater than 3.5.[2]
Treatment
Atherosclerotic renal artery stenosis
It is initially treated with medications, including diuretics, and medications for blood pressure control.[13] When high-grade renal artery stenosis is documented and blood pressure cannot be controlled with medication, or if renal function deteriorates, surgery may be resorted to. The most commonly used procedure is a minimally-invasive angioplasty with or without stenting. Essentially, stenting is a procedure in which stents (tiny, expandable mesh tubes) are placed to hold open an artery and potentially improve blood flow. It is unclear if this approach yields better results than the use of medications alone.[22] It is a relatively safe procedure.[22] If all else fails and the kidney is thought to be worsening hypertension and revascularization with angioplasty or surgery does not work, then surgical removal of the affected kidney (nephrectomy) may significantly improve high blood pressure.[23]
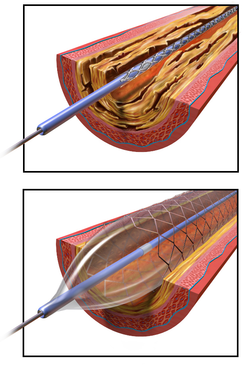
Stenting vs. medical therapy
Historically, balloon dilation of vascular lesions was first performed in 1964. The first treatment of renovascular lesions by percutaneous catheter angioplasty (PTA) was performed in 1978 by Andreas Roland Grüntzig. Stenting became the standard of endovascular care for ostial stenoses because of the poor technical success of PTA.[24] The use of renal artery stents, particularly for ostial lesions, has further improved long-term patency rates.[24] Further research into the efficacy of renal artery stenting has since been conducted, including the ASTRAL and CORAL trials, and research is ongoing as to whether there is a clear benefit to stenting over more conservative management, such as by medications alone.
The ASTRAL trial [25] randomized 806 patients to renal artery revascularization via stenting versus medical therapy, with the following results:
- No survival advantage in the stented group
- No benefit for renal function in the stented group
- Serious complications with stenting (2 deaths, 3 amputations, others)
The CORAL study [26] randomized 947 patients to stenting versus medical therapy, with a primary endpoint of major cardiovascular or renal events. Similarly, this study also found there was no benefit to stenting over medical treatment alone.
Collectively, the evidence so far does not support renal artery stenting as a proper mainstay treatment for stenosis because it conveys no survival benefit, no true renoprotection, and does not decrease the need for antihypertensive medications. A debate remains about whether or not it is beneficial to stent for purely ostial lesions, and comparing the success in treating with angioplasty (such as balloon dilatation) versus stenting. Smoking cessation and thoughtful blood pressure control, via lifestyle/dietary changes and medication, are the current mainstays of managing renovascular hypertension.
Fibromuscular dysplasia
Angioplasty alone is preferred in fibromuscular dysplasia, with stenting reserved for unsuccessful angioplasty or complications such as dissection.[27]
Epidemiology and risk factors
Renal artery stenosis (RAS) is a common cause of secondary hypertension (HTN), affecting <1% of patients with mild HTN but up to 10%–40% of those with severe or refractory HTN. As stated previously, RAS is predominantly caused by atherosclerosis, which is more common in older patients. In younger patients, especially women, fibromuscular dysplasia (FMD) can be attributed to a 75%–80% incidence of RAS. RAS is found in roughly 7% of elderly patients and is responsible for 5–22% of chronic kidney disease (CKD) in patients older than 50. Prevalence is higher in patients with other vascular diseases, including those with peripheral vascular disease and/or those requiring coronary catheterization. [28][29]
Risk factors strongly associated with atherosclerotic RAS include HTN, age (>60), sex (male), smoking, diabetes, and cardiovascular diseases, such as PVD and a history of myocardial infarction. Whereas FMD-related RAS is more common in females (~90% of cases).[30][31]
Prognosis
Patients with total occlusion or high-grade stenoses (e.g., >95%) of one or more renal arteries have an approximately 50% four-year survival rate. [32] The more acute causes of death are usually related to cardiovascular disease, including heart attacks, stroke, and heart failure, often driven by severe and resistant hypertension. Over the long term, death may be caused by chronic renal failure, which is associated with fluid overload, electrolyte abnormalities, and the accumulation of urea, creatinine, and acidic compounds in the blood (uremia).[33][34]
References
- ^ a b c “Renal Artery Stenosis”. National Institute of Diabetes and Digestive and Kidney Diseases. Archived from the original on 16 December 2016. Retrieved 17 August 2015.
- ^ a b c Granata A, Fiorini F, Andrulli S, Logias F, Gallieni M, Romano G, et al. (December 2009). “Doppler ultrasound and renal artery stenosis: An overview”. Journal of Ultrasound. 12 (4): 133–143. doi:10.1016/j.jus.2009.09.006. PMC 3567456. PMID 23397022.
- ^ a b Ong YY (1 January 2005). A Clinical Approach to Medicine. World Scientific. ISBN 978-981-256-073-5. Archived from the original on 4 September 2024. Retrieved 28 October 2020.
- ^ “Renal artery stenosis – Symptoms and causes”. Mayo Clinic. Retrieved 12 March 2026.
- ^ “UpToDate”. www.uptodate.com. Retrieved 12 March 2026.
- ^ “Renal Artery Stenosis | Boston Children’s Hospital”. www.childrenshospital.org. Retrieved 12 March 2026.
- ^ MedlinePlus Encyclopedia: Renovascular hypertension
- ^ Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR, et al. (November 2018). “Resistant Hypertension: Detection, Evaluation, and Management: A Scientific Statement From the American Heart Association”. Hypertension. 72 (5). doi:10.1161/HYP.0000000000000084. ISSN 0194-911X. PMC 6530990. PMID 30354828.
- ^ Messerli FH, Bangalore S, Makani H, Rimoldi SF, Allemann Y, White CJ, et al. (2 September 2011). “Flash pulmonary oedema and bilateral renal artery stenosis: the Pickering Syndrome”. European Heart Journal. 32 (18): 2231–2235. doi:10.1093/eurheartj/ehr056. PMID 21406441.
- ^ “Flash Pulmonary Edema – an overview | ScienceDirect Topics”. www.sciencedirect.com. Retrieved 17 March 2026.
- ^ Vassallo D, Kalra PA (2017). “Atherosclerotic renovascular disease – epidemiology, treatment and current challenges”. Advances in Interventional Cardiology. 3 (3): 191–201. doi:10.5114/aic.2017.70186. PMC 5644037. PMID 29056991.
- ^ Safian RD (March 2021). “Renal artery stenosis”. Progress in Cardiovascular Diseases. 65: 60–70. doi:10.1016/j.pcad.2021.03.003. PMID 33745915. S2CID 232311595.
- ^ a b c Renal Artery Stenosis at eMedicine
- ^ Steyerberg E (16 December 2008). Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating. Springer Science & Business Media. ISBN 978-0-387-77244-8. Archived from the original on 4 September 2024. Retrieved 28 October 2020.
- ^ “Renal artery stenosis – Diagnosis and treatment – Mayo Clinic”. www.mayoclinic.org. Retrieved 26 March 2026.
- ^ Mehdi A, Taliercio JJ, Nakhoul G (1 November 2020). “Contrast media in patients with kidney disease: An update”. Cleveland Clinic Journal of Medicine. 87 (11): 683–694. doi:10.3949/ccjm.87a.20015. ISSN 0891-1150. PMID 33139262.
- ^ Protasiewicz M, Kądziela J, Początek K, Poręba R, Podgórski M, Derkacz A, et al. (November 2013). “Renal Artery Stenosis in Patients With Resistant Hypertension”. The American Journal of Cardiology. 112 (9): 1417–1420. doi:10.1016/j.amjcard.2013.06.030. PMID 24135303.
- ^ Talley NJ, O’Connor S (20 September 2013). Clinical Examination: A Systematic Guide to Physical Diagnosis. Elsevier Health Sciences. ISBN 978-0-7295-4147-3. Archived from the original on 4 September 2024. Retrieved 28 October 2020.
- ^ Taylor AT (May 2014). “Radionuclides in Nephrourology, Part 2: Pitfalls and Diagnostic Applications”. Journal of Nuclear Medicine. 55 (5): 786–798. doi:10.2967/jnumed.113.133454. PMC 4451959. PMID 24591488.
- ^ Sam AH, James T.H. Teo (2010). Rapid Medicine. Wiley-Blackwell. ISBN 978-1-4051-8323-9.
- ^ Attenberger UI, Morelli JN, Schoenberg SO, Michaely HJ (December 2011). “Assessment of the kidneys: magnetic resonance angiography, perfusion and diffusion”. Journal of Cardiovascular Magnetic Resonance. 13 (1): 70. doi:10.1186/1532-429X-13-70. PMC 3228749. PMID 22085467.
- ^ a b Jenks S, Yeoh SE, Conway BR (5 December 2014). “Balloon angioplasty, with and without stenting, versus medical therapy for hypertensive patients with renal artery stenosis”. Cochrane Database of Systematic Reviews. 2014 (12) CD002944. doi:10.1002/14651858.CD002944.pub2. PMC 7138037. PMID 25478936.
- ^ Fine RN, Webber SA, Harmon WE, Kelly D, Olthoff KM (8 April 2009). Pediatric Solid Organ Transplantation. John Wiley & Sons. ISBN 978-1-4443-1273-7. Archived from the original on 4 September 2024. Retrieved 28 October 2020.
- ^ a b Bush RL, Najibi S, MacDonald MJ, Lin PH, Chaikof EL, Martin LG, et al. (May 2001). “Endovascular revascularization of renal artery stenosis: technical and clinical results”. Journal of Vascular Surgery. 33 (5): 1041–1049. doi:10.1067/mva.2001.111486. ISSN 0741-5214. PMID 11331847.
- ^ null n (12 November 2009). “Revascularization versus Medical Therapy for Renal-Artery Stenosis”. New England Journal of Medicine. 361 (20): 1953–1962. doi:10.1056/NEJMoa0905368. hdl:2381/17521. ISSN 0028-4793. PMID 19907042.
- ^ Cooper CJ, Murphy TP, Cutlip DE, Jamerson K, Henrich W, Reid DM, et al. (2 January 2014). “Stenting and Medical Therapy for Atherosclerotic Renal-Artery Stenosis”. New England Journal of Medicine. 370 (1): 13–22. doi:10.1056/NEJMoa1310753. ISSN 0028-4793. PMC 4815927. PMID 24245566.
- ^ Chrysant SG, Chrysant GS (February 2014). “Treatment of hypertension in patients with renal artery stenosis due to fibromuscular dysplasia of the renal arteries”. Cardiovascular Diagnosis and Therapy. 4 (1): 36–43. doi:10.3978/j.issn.2223-3652.2014.02.01. PMC 3943779. PMID 24649423.
- ^ Rout P, Rauf N, Bokhari SR (2026), “Renal Artery Stenosis”, StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 28613469, retrieved 26 March 2026
- ^ “UpToDate”. www.uptodate.com. Retrieved 26 March 2026.
- ^ “UpToDate”. www.uptodate.com. Retrieved 26 March 2026.
- ^ “UpToDate”. www.uptodate.com. Retrieved 26 March 2026.
- ^ Annest S. “Renal Artery Stenosis: Is It Common & Life Threatening?”.
{{cite web}}: CS1 maint: url-status (link) - ^ Liabeuf S, Pepin M, Franssen CF, Viggiano D, Carriazo S, Gansevoort RT, et al. (28 December 2021). “Chronic kidney disease and neurological disorders: are uraemic toxins the missing piece of the puzzle?”. Nephrology, Dialysis, Transplantation: Official Publication of the European Dialysis and Transplant Association – European Renal Association. 37 (Suppl 2): ii33–ii44. doi:10.1093/ndt/gfab223. ISSN 1460-2385. PMC 8713157. PMID 34718753.
- ^ CDC (13 August 2025). “Chronic Kidney Disease in the United States, 2023”. Chronic Kidney Disease. Retrieved 26 March 2026.
Further reading
- Schrier RW (1 January 2010). Renal and Electrolyte Disorders. Lippincott Williams & Wilkins. ISBN 978-1-60831-072-2.